|
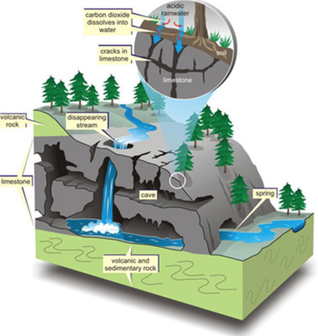
This week's science topic of caves was suggested by one of the students for us. Caves are formed when are usually made when water runs over soft rock, such as limestone. The acid in the water slowly eats away the limestone, making a hole. The hole gets larger and larger. If the water finds a new path, the cave is left dry.
As you can see in the photos there were a few caves beginning to form in the cups where the sugar was washed away by the 'rain'. However, as can happen sometimes in science experiments, it doesn't always work out exactly as planned!
As mentioned above if the water finds a new path out the cave is left dry. Unfortunately in our caves the holes made at the bottom weren't big enough and our caves were definitely underwater caves. But we learnt the cave formation process and scientists learn both from observations and mistakes in their experiments. And we had a lot of fun with the messiness as well.
0 Comments
In preparation for learning how caves are formed we learnt about the types of rocks today. Although there are many types of rocks on earth and we can name lots of them, we learnt that they can all categorised as three main types: -Igneous -Sedimentary -Metamorphic As we only have one lunch time to learn about something you could study for years we kept it very simple. And also used food to help us understand the types. Igneous rocks are formed when magma (molten rock deep within the earth) cools and hardens. Sedimentary rocks are formed from particles of sand, shells, pebbles, and other fragments of materials. Over many hundreds of thousands of years these layers of sediments form on top of each other. We made our own sedimentary rocks out of layers of bread, rice bubbles, chocolate chips and coconut to represent the layers. Metamorphic rocks are formed when the original rock changes (metamorphosis) from either intense heat or pressure. We chose to change our rocks with pressure and squashed our rocks down. And of course we got to eat our rocks at the end!!! Here is a video to help you learn more about rocks at home.
For our first experiment of the year we tested our hypothesising skills and observations skills. First we observed what happened when we mixed two jars of red and blue cold water together. We observed that they mixed quickly to become purple. We then thought about what would happen if one of the jars was hot water. The photos show some of the things we observed: In term two we start up science club at school. On a Wednesday lunchtime we gather in Room 1 with Miss Schollum to learn to hypothesise, observe, record and learn about the world around us.
If you This may just be the easiest, messiest, and most fun science activity I know. It is a classic, and I have gotten several requests recently to post directions. You should know that if you try this activity and you are not smiling and messy with corn starch goo at the end, then you are definitely doing something wrong. Also keep in mind that this is not just about fun, there is some pretty amazing science going on here. You will need:
Why does my Gloop act like that? Your Gloop is made up of tiny, solid particles of cornstarch suspended in water. Chemists call this type of mixture a colloid. As you found out when you experimented with your Gloop, this colloid behaves strangely. When you bang on it with a spoon or quickly squeeze a handful of Gloop, it freezes in place, acting like a solid. The harder you push, the thicker the Gloop becomes. But when you open your hand and let your Gloop ooze, it drips like a liquid. Try to stir the Gloop quickly with a finger, and it will resist your movement. Stir it slowly, and it will flow around your finger easily. Smack water with a spoon and it splashes. Smack Gloop with a spoon and it acts like a solid. Most liquids don't act like that. If you stir a cup of water with your finger, the water moves out of the way easily--and it doesn't matter whether you stir it quickly or slowly. Your finger is applying what a physicist would call a sideways shearing force to the water. In response, the water shears, or moves out of the way. The behavior of Gloop relates to its viscosity, or resistance to flow. Water's viscosity doesn't change when you apply a shearing force--but the viscosity of your Ooze does. Back in the 1700s, Isaac Newton identified the properties of an ideal liquid. Water and other liquids that have the properties that Newton identifies are call Newtonian fluids. Your Gloop doesn't act like Newton's ideal fluid. It's a non-Newtonian fluid. There are many non-Newtonian fluids around. They don't all behave like your Gloop, but each one is weird in its own way. Ketchup, for example, is a non-Newtonian fluid. (The scientific term for this type of non-Newtonian fluid is thixotropic. That comes from the Greek words thixis, which means "the act of handling" and trope, meaning "change".) Quicksand is a non-Newtonian fluid that acts more like your Gloop--it gets more viscous when you apply a shearing force. If you ever find yourself sinking in a pool of quicksand (or a vat of cornstarch and water), try swimming toward the shore very slowly. The slower you move, the less the quicksand or cornstarch will resist your movement. Scientific concept: When scientists do experiments where they have to change variables it is always important to have a control - the original that you can compare your new models to. If you don't have a control how will you know if the changes you make or for the better or actually making it slower or weaker? The other thing we looked at was only changing one thing at a time. If we change lots of things all together how will we know if they are all for the best?
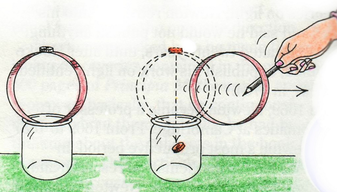
To help support this concept of having a control we made a helicopter out of paper and labelled it as our control. Once we had our control made we could start changing things on the original to try and make it faster or slower. We looked at changing the blades, the weights with the paperclips and the length of the base to see how this changed the descension. After making many variables we had a cmpetition at the end to see who had made the fastest and slowest helicopters. Scientific concept: Newton's first law states 'an object at rest will remain at rest unless acted upon by an outside force'. To help us understand this law we started by looking at the diver experiment. When a coin is put on top of a cardboard loop on a jar and the loop is taken away, what happens to the coin? Some students thought it would land on the table, others in the jar and some thought it would stay on the paper. When the loop was taken away we could observe that the coin dropped straight down. This is what Newton's law tell us - the hoop moved so fast that the coin didn't have the friction of an outside force acting on it so it went straight down. To help us understand this more and learn hands on we made our own inertia towers where we could take pieces of card away quickly so that the blocks and cups didn't feel the outside force of movement. Well done to Lakia who managed a tower of 5 blocks still standing after pulling away the card! Scientific concept: When you look at a drop of water it makes a dome shape rather than run flat on a table. This is caused by water surface tension. Surface tension is created when the water molecules hold on tightly to each other. The molecules at the surface hold on to each other even more because there is no water molecule above them to grab on to. This tension keeps the drop of water together in the dome shape. You may have seen drops of water on leaves and observed the domes of water they formed, but have you ever thought how much water could fit on before it all falls off? This week we first investigated the shapes and nature of drops of water and how we can break the water tension. We then came back together to share our observations. Part of being a scientist is to make predictions and then conduct an experiment to see if our predictions are correct. Based on our observations we all made a prediction of how many drops of water we could fit on a 10 cent coin. Our predictions ranged from 5-10 drops. Through multiple testings and changing the variables, such as turning the coin over, mean that we were confident in our observations. The most drops some of us put on the coin was 32! A lot more than any of us predicted. Scientific concept: When you rub a balloon on wool or your hair it creates static electricity. Static electricity is negatively charged particles (called electrons) jumping over to positively charged objects. When you rub the balloon against your hair (the most fun option) it becomes negatively charged. It has taken some of the electrons from the hair and left them positively charged. The positively charged hair is attracted to the negatively charged balloon and starts to rise up to meet it. To show how static electricity works we tried out a variety of experiments. We got some balloons and rubbed them on our jumpers, the carpet and hair to negatively charge them. We then tried to attract a variety of items such as tissue paper, straws, cans and salt and pepper. We also observed during our experiments that some students were great conductors of electricity! Scientific Concept:A bubble is just air wrapped in soap film. Soap film is made from soap and water (or other liquid). The outside and inside surfaces of a bubble consist of soap molecules. A thin layer of water lies between the two layers of soap molecules, sort of like a water sandwich with soap molecules for bread. They work together to hold air inside. For the first science club activity we looked bubbles and how we could blow the best bubble. After discussing what we knew about making bubble mixture and the scientific concept behind bubbles, we got to all have a turn blowing bubbles. We each had a pipette with the end cut off to blow through and tried to work out the best way to blow a bubble. We also experimented with what happened when we added more dishwashing liquid to the solution. We then came back together to think about what would happen if we added glycerol to the solution. Ask your child what they observed! |
On WEDNESDAY lunchtimes Year 2-6 students come together to learn about scientific concepts. Archives
November 2017
|
| St Albans' Science Club |
|
 RSS Feed
RSS Feed
